
It also looked at the overall patient experience with the next-generation system. Medtronic presented one- and three-month outcomes from an evaluation of the long-term performance of its closed-loop SCS system. “We are committed to advancing the field of neuromodulation through rigorous scientific research in order to deliver on our Mission to alleviate pain, restore health and extend life to patients around the world.” Closed-loop SCS meets primary objective 
“Insights gained today from new clinical evidence pave the way toward the revolutionary therapies of tomorrow, and Medtronic is proud to be leading the way at NANS 2024 with 26 accepted clinical abstracts,” said Paolo Di Vincenzo, president of the Neuromodulation business at Medtronic. and Australia.Īdditionally, Medtronic shared 24-month outcomes for its DTM SCS compared to conventional medical management (CMM). The system holds European and Japanese approval but remains investigational in the U.S. That study looked at the company’s investigational, next-generation rechargeable SCS system. Medtronic’s data included evaluations of closed-loop SCS over three months in Australia. The company also plans to highlight its recently FDA-approved Percept RC deep brain stimulation system at NANS. Results demonstrated the benefits of SCS in patients with chronic low back pain (CLBP) and leg pain. 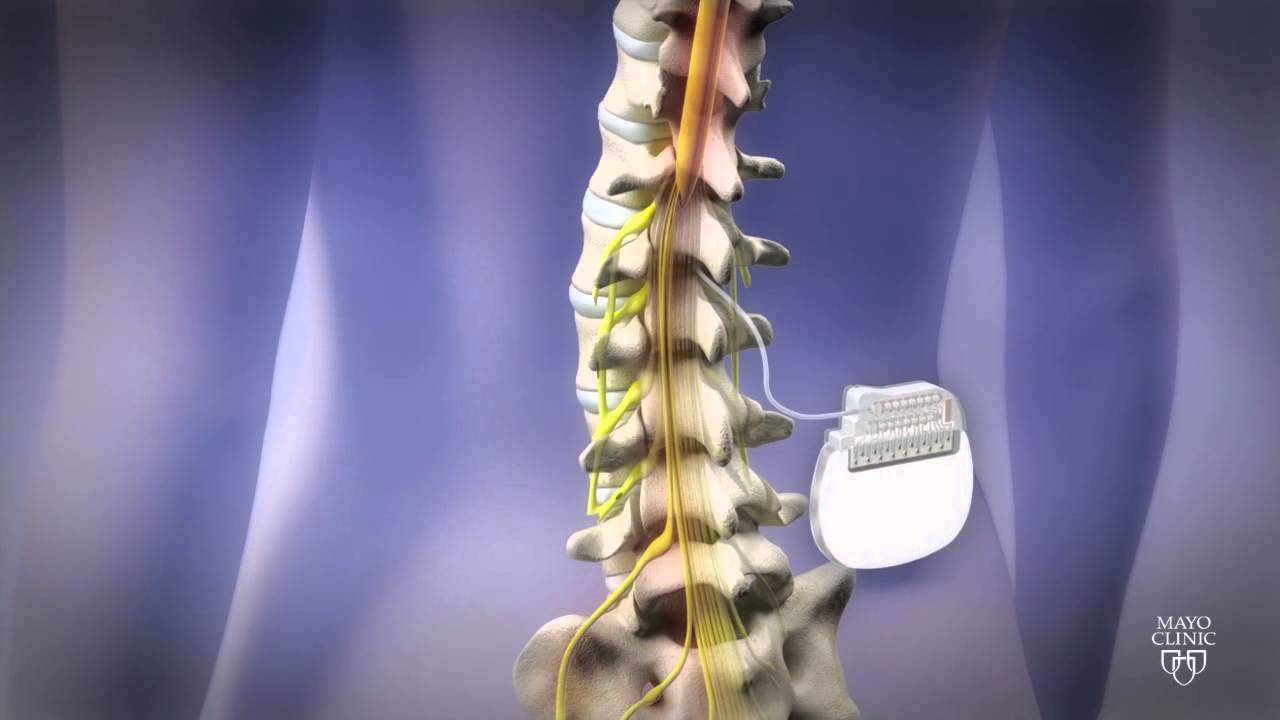
The medtech giant shared data at the North American Neuromodulation Society (NANS) annual meeting in Las Vegas. + today announced data from two clinical trials supporting its spinal cord stimulation (SCS) technologies. The Vanta recharge-free neurostimulator offers DTM therapy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
